
Direct measurements for global temperature only date back to about 1850, so how do we figure out what past climates were like?
Once we move beyond the limits of direct measurements, we need to use proxies—preserved physical characteristics of past environments. Oxygen isotopes are the most commonly used paleoproxy to reconstruct long records of past climates.
Out of the three stable isotopes of oxygen, two are typically used in paleoclimate studies, 16O and 18O. The lighter 16O accounts for the vast majority of total oxygen—99.8% of it—while the heavier 18O accounts for most of the small remainder.
Water on Earth can therefore have H2O with a 16O or with an 18O. Due to the fact that 16O is lighter and has a lower bond energy, it will evaporate more readily than 18O. Similarly, because 18O is heavier and has a higher bond energy, it will preferentially remain in liquid form. This difference in mass and chemical properties leads to isotope fractionation—meaning that the relative abundance of 18O to 16O can change in a system. The degree of fractionation is temperature dependent, and cooler temperatures increase fractionation.
So how do we apply this isotopic fractionation to understanding past climates? Take an ocean with a set reservoir of 18O and 16O. Water with 16O will preferentially evaporate, which then condenses into cloud vapor. When these clouds produce rain, water with 18O will preferentially be rained out. As you remove this heavier 18O through precipitation, the resulting vapor and liquid will become lighter, meaning there is a higher relative abundance of 16O to 18O. Once these clouds reach the poles, the water will become locked in ice. The ice is thus enriched in 16O and is isotopically light. Conversely, the water in the oceans are now enriched in 18O and are isotopically heavy, because you preferentially removed 16O and locked it away in ice.
Now, imagine if you have no ice sheets. Instead of the 16O becoming locked in ice, it will cycle back to the ocean so that your oceans are no longer isotopically heavy.
Because the degree of fractionation is temperature dependent, this type of fractionation will be increased at colder temperatures. Oxygen isotopes will therefore be representative of both temperature and ice volume.
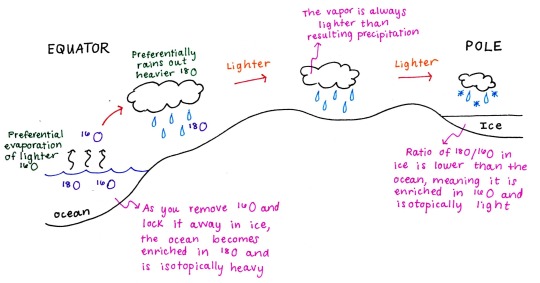
Scientists can take oxygen isotope measurements from ice cores or from deep sea sediment cores to reconstruct climate. If you measure heavy oxygen isotope values enriched in 18O in deep sea sediments (representative of ocean isotopic composition), that would be interpreted to indicate that the climate was cooler. Colder temperatures produce greater fractionation effects and you can also have water being trapped in polar ice. Ice cores from this same interval would show the opposite isotopic signal, because they are enriched in light 16O.
Conversely, deep sea sediments that are not enriched in 18O would indicate a warmer climate, because you have less fractionation and wouldn’t have lighter 16O locked in polar ice. Oxygen isotope records from deep sea sediments have been used to reconstruct past climate over the Cenozoic (the last 65 million years). Isotope values show that climate overall has been cooling for the last 50 million years.
